Helus Pharma Announces Topline Results in Phase 2 Signal Detection Study for HLP004 in Patients with
- Statistically significant (p<0.0001) and clinically meaningful improvement in Hamilton Anxiety Rating Scale (“HAM-A”) of ~10 points on top of Standard of Care (“SoC”) at 6 weeks
- Durable effects sustained through at least 6 months
- In Phase 1 trial most participants were ready for discharge within 3 hours1; Acute effects lasted ~90 minutes
- Generally well-tolerated, adverse events were transient, with no drug related serious adverse events recorded
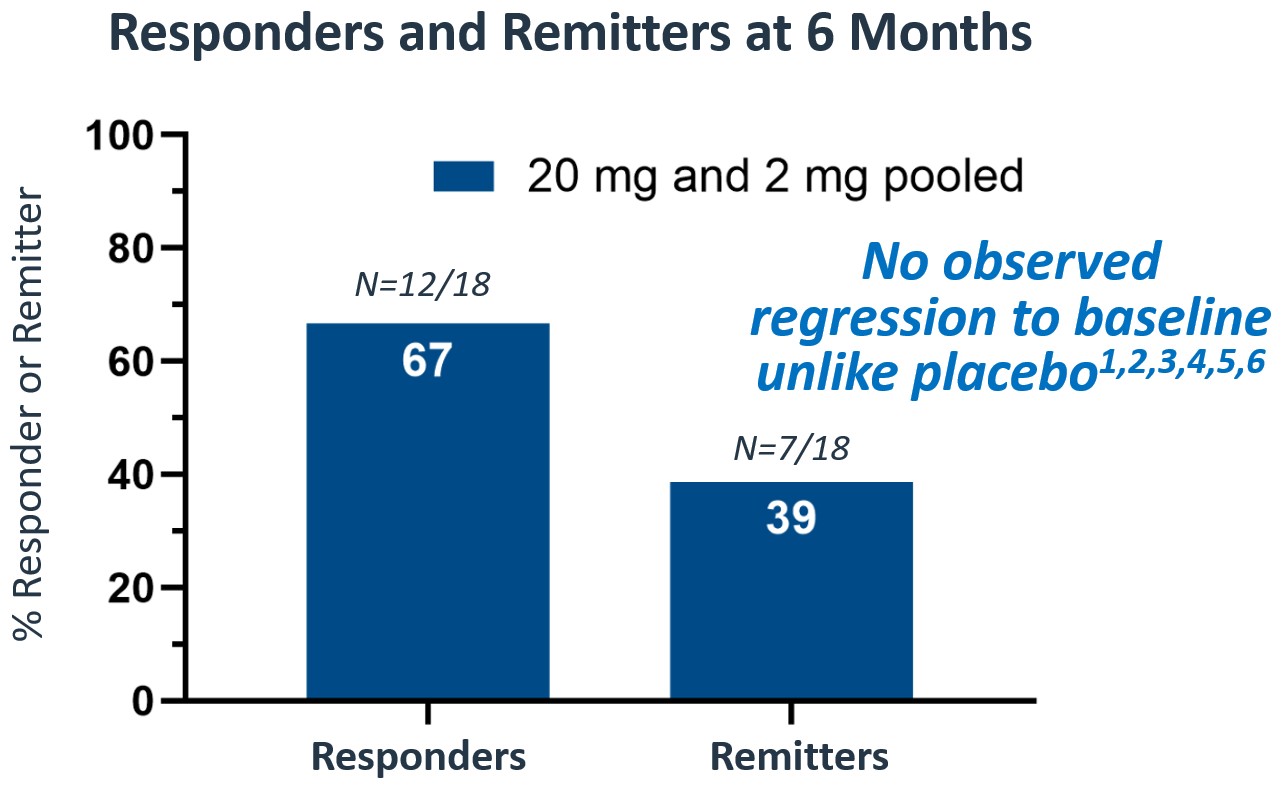
- At six months, the pooled study population showed 67% responders and 39% of patients were in remission (see Figure 1 below)
Figure 1
This news release constitutes a "designated news release" for the purpose of the Company's prospectus supplement dated December 30, 2025, to its short form base shelf prospectus dated September 17, 2025, as amended on December 19, 2025.
NEW YORK and TORONTO, March 05, 2026 (GLOBE NEWSWIRE) -- Helus Pharma (Nasdaq: HELP; Cboe CA: HELP), a clinical-stage pharmaceutical company developing novel serotonergic agonists (“NSAs”) for serious mental health conditions, today announced topline results from a Phase 2 signal detection study evaluating HLP004 as a potential treatment for adults with moderate-to-severe generalized anxiety disorder (“GAD”) who remained symptomatic despite ongoing SoC antidepressant therapy, including selective serotonin reuptake inhibitors and related agents.
GAD affects more than 20 million adults in the United States, and approximately half of patients treated for GAD fail to respond to initial first-line therapy.2,3 No adjunctive pharmacologic treatment for GAD has ever been approved. No new monotherapy has been approved in almost two decades.
In the Phase 2 signal detection study, 36 patients were randomized 2-to-1 active-to-placebo to HLP004 20 mg or 2mg and received two intramuscular doses three weeks apart. Patients were followed through Week 12, with continued observational follow-up extending up to one year. Participants had an average baseline HAM-A score of 22 and a General Anxiety Disorder-7 score of greater than or equal to 10 at screening.
All study participants were already being treated, and continued treatment throughout the trial, with SoC medications for generalized anxiety disorder. The 10-point improvement in anxiety symptoms is above and beyond what was already being seen with SoC treatment.
Key findings include:
- Clinically meaningful efficacy: Patients that received 20mg HLP004 adjunctive to SoC therapy achieved mean reduction of 10.4-points (p<0.0001) in the HAM-A from baseline at six weeks.
- Efficacy in difficult to treat population: Study population consisted of moderate-to-severe patients who remained symptomatic despite ongoing antidepressant or anxiolytic therapy.
- Durable remission and robust response over time:
- At six months, the pooled study population showed 67% responders and 39% remitters.
- Participants randomized to both 20 mg and 2mg dosing arms experienced meaningful subjective effects and showed clinically significant responses over SoC, with 59% meeting the criteria for response and 32% for remission in the 20mg arm and a 30% responder and remitter rate in the 2mg arm at week 6.
- Commercially scalable clinic time: Short in-clinic treatment experience with acute drug effects lasting approximately 90 minutes and discharge readiness within approximately three hours1, fitting within the treatment paradigm of existing interventional psychiatry clinics.
- Well tolerated: Favorable tolerability profile with no drug-related serious adverse events or suicidality-related safety signals.
“These Phase 2 results support the continued development of HLP004, and I am encouraged by the magnitude of improvement observed over standard of care treatments, together with the rapid onset and short in-clinic treatment experience for this patient population with limited options,” noted Dr. Andrew Cutler, Clinical Professor of Psychiatry at SUNY Upstate Medical University and Senior Advisor to Helus Pharma.
Michael Cola, Chief Executive Officer of Helus Pharma, said, “Patients living with generalized anxiety disorder remain significantly underserved, with many continuing to struggle despite currently available treatments. We are encouraged by these data and the potential for HLP004 to bring hope to GAD patients. The Company’s broad intellectual property portfolio has been leveraged once again to create what we feel are best in class products and we are further excited to release the data on HLP003 targeted at major depressive disorder in the fourth quarter of 2026.”
For additional information, please access our webcast, which is available on the Company's investor relations website on the Events and Presentations page.
About HLP004
HLP004 is an investigational intramuscular deuterated serotonergic agonist designed to activate serotonin pathways believed to promote neuroplasticity. The program is being developed as a potential adjunctive treatment for patients with generalized anxiety disorder who remain symptomatic despite existing pharmacologic therapies.
About Helus Pharma
Helus Pharma™, the commercial operating name of Cybin Inc. (the “Company” or “Helus Pharma”) is a clinical stage pharmaceutical company committed to helping minds heal by developing proprietary NSAs - novel serotonergic agonists: synthetic molecules designed to activate serotonin pathways that are believed to promote neuroplasticity. The Company’s proprietary NSAs are intended to address the large unmet need for people who suffer from depression, anxiety, and other mental health conditions.
With class leading data, Helus Pharma aims to improve the treatment landscape through the introduction of NSAs that aim to provide durable improvements in mental health. Helus Pharma is currently developing HLP003, a proprietary NSA, in Phase 3 clinical development for the adjunctive treatment of major depressive disorder that has received Breakthrough Therapy Designation from the U.S. Food and Drug Administration and HLP004, also a proprietary NSA in Phase 2 for generalized anxiety disorder. Additionally, Helus Pharma has an extensive research portfolio of investigational NSAs.
The Company operates in Canada, the United States, the United Kingdom, and Ireland. For Company updates and to learn more about Helus Pharma, visit www.helus.com or follow the team on X, LinkedIn, YouTube and Instagram. Helus PharmaTM is a trademark of Cybin Corp.
Notes
1. In Phase 1 study at 30 mg dose.
2. Ringeisen H, et al. Mental and Substance Use Disorders Prevalence Study: Findings Report. RTI International; 2023.
3. Fagan H, et al. Pharmacological Treatment of Generalized Anxiety Disorder: Current Practice and Future Directions. Expert Review of Neurotherapeutics. 2023.
Figure 1 Notes:
1) Gueorguieva, et.al. Lancet Psychiatry, 2017. 4(3):230-237.
2) Rutherford, et al. Depress Anxiety, 2015. 32(12):944-57.
3) Berwian, I.M., et al. Psychol Med, 2017. 47(3):426-437.
4) Jones, B.D.M., et al.JAMA Network Open, 2021. 4(9):e2125531-e2125531.
5) Davidson, et al. Eur Neuropsychopharmacol, 2008. 18(9):p. 673-81.
6) Quitkin, et al. Arch GenPsychiatry, 1984. 41:p. 782-786.
Cautionary Notes and Forward-Looking Statements
Certain statements in this news release relating to the Company are forward-looking statements or forward-looking information within the meaning of applicable securities laws (collectively, “forward-looking statements”) and are prospective in nature. Forward-looking statements are not based on historical facts, but rather on current expectations and projections about future events and are therefore subject to risks and uncertainties which could cause actual results to differ materially from the future results expressed or implied by the forward-looking statements. These statements generally can be identified by the use of forward-looking words such as “may”, “should”, “could”, “potential”, “possible”, “intend”, “estimate”, “plan”, “anticipate”, “expect”, “believe” or “continue”, or the negative thereof or similar variations. Forward-looking statements in this news release include statements regarding the Company’s expectations of progressing the HLP004 program toward development; the potential of HLP004 as a scalable treatment option for patients living with GAD; and the Company’s plans to engineer proprietary drug discovery platforms, innovative drug delivery systems, novel formulation approaches and treatment regimens for mental health conditions.
These forward-looking statements are based on reasonable assumptions and estimates of management of the Company at the time such statements were made. Actual future results may differ materially as forward-looking statements involve known and unknown risks, uncertainties, and other factors which may cause the actual results, performance, or achievements of the Company to materially differ from any future results, performance, or achievements expressed or implied by such forward-looking statements. Such factors, among other things, include: fluctuations in general macroeconomic conditions; fluctuations in securities markets; expectations regarding the size of the NSA market; the ability of the Company to successfully achieve its business objectives; plans for growth; political, social and environmental uncertainties; employee relations; the presence of laws and regulations that may impose restrictions in the markets where the Company operates; implications of disease outbreaks on the Company's operations; and the risk factors set out in each of the Company's management's discussion and analysis for the three and nine month periods ended December 31, 2025, and the Company’s annual information form for the year ended March 31, 2025, which are available under the Company's profile on SEDAR+ at www.sedarplus.ca and with the U.S. Securities and Exchange Commission on EDGAR at www.sec.gov/edgar. Although the forward-looking statements contained in this news release are based upon what management of the Company believes, or believed at the time, to be reasonable assumptions, the Company cannot assure shareholders that actual results will be consistent with such forward-looking statements, as there may be other factors that cause results not to be as anticipated, estimated or intended. Readers should not place undue reliance on the forward-looking statements contained in this news release. The Company assumes no obligation to update the forward-looking statements of beliefs, opinions, projections, or other factors, should they change, except as required by law.
The Company makes no medical, treatment or health benefit claims about the Company’s proposed products. The U.S. Food and Drug Administration, Health Canada or other similar regulatory authorities have not evaluated claims regarding NSAs or HLP003, HLP004 and other programs of the Company. The efficacy of such products has not been confirmed by approved research. There is no assurance that the use of NSAs, HLP003, HLP004 or other programs of the Company can diagnose, treat, cure or prevent any disease or condition. Rigorous scientific research and clinical trials are needed. If Helus Pharma cannot obtain the approvals or research necessary to commercialize its business, it may have a material adverse effect on the Company’s performance and operations.
Neither Cboe Canada, nor the Nasdaq Global Market stock exchange, have approved or disapproved the contents of this news release and are not responsible for the adequacy and accuracy of the contents herein.
Investor Contact:
Josh Barer
astr partners
Managing Director
(908) 578-6478
josh.barer@astrpartners.com
George Tziras
Chief Business Officer
Helus Pharma
1-866-292-4601
irteam@helus.com – or – media@helus.com
Media Contact:
Johnny Tokarczyk
RXMD
Public Relations Director
jtokarczyk@rxmedyn.com
(914) 772-7562
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/728bf286-24d0-40b0-a151-beaced3735d7
- 新年伊始,慧科讯业邀您共同用36+份报告回顾2025
- 女性用户在‘萝卜快跑’车内体验护肤新方式
- NetApp任命Paul Fipps为董事会成员
- 春日焕新好时节,顾家家居春季发布会推出多款重磅新品
- 2024 LSCM物流高峰会 探讨大灣區智慧城市及数字经济发展 助业界把握机遇
- 中国移动江苏公司走进“苏.皋长寿”年货大集共赴暖皋之约
- 易事特三大智造基地全速冲刺,点燃GWh级大型储能项目交付热潮
- 探秘百山祖黑松露酱价差真相:原料品质与含量决定价格高低
- NK细胞疗法破局实体瘤:广州赛奥金ZMPB‑NK006(国认唯一)—— 现货型 1 小时快速干预中晚期实体瘤
- 美聚缘酒行——引领品质生活的电商新选择
- 首届“营动中国”全国青少年户外运动大赛成功举行
- View韩国地包天手术前自测:三种地包天类型,你属于哪一种?
- 以升能源Yinson Renewables:走进这家来自马来西亚的绿色巨擘
- 豆神教育、智谱AI等成立智创科技公司,注册资本5亿元
- 摩飞MR2156肩颈按摩仪专业推拿手法解压好物
- 2025十大放心猫粮国产排行榜,守护猫咪健康成长
- 瑞派明星专科:攻克骨疾难题,撑起健康“生命架构”
- arena阿瑞娜迎来品牌代言人费立纬,携手泳者新一代
- 中国黑莓采摘节发起者——悠果维引领健康食品新风尚
- 孩子总是抱怨看书眼睛累,是不爱学习的借口?可能是一眼清晰一眼模糊所致!——福州爱尔眼科
- 奶奶青光眼急发,陪诊的孙女意外终结了十余年斜眼看人的尴尬,爷爷也……
- 易事特等企业联合攻坚“储能型钠离子电池及其关键材料研发及产业化”等重点项目
- 八载同行,创新不止:嘉吉携全链路解决方案亮相第八届进博会
- 彰显中国榜样 时代楷模江苏宏碧能源科技集团有限公司董事长—— 蔡成建
- 首届智慧照明博览燃亮创新照明的未来
- 看得见的父爱 摸得着的真情——读于海洋先生新作《父亲》有感
- 深耕功能农业 赋能健康生活——安徽田园志助力国民营养升级
- “研发在东部 生产在呼市”全国联动新模式呼和浩特布局合成生物产业
- “退役军人医疗救助”项目捐赠暨“2024退役军人家庭-全年医疗费用保险卡”升级上线启动仪式在京举行
- 壮乡骄子温德里格:文武双全,以法律为桥连通世界——记广西超冠律师事务所高级合伙人、国际律师的文化法治之路
推荐
-
 看新东方创始人俞敏洪如何回应董宇辉新号分流的?
(来源:中国证券报)
东方甄选净利润大幅下滑
资讯
看新东方创始人俞敏洪如何回应董宇辉新号分流的?
(来源:中国证券报)
东方甄选净利润大幅下滑
资讯
-
 奥运冠军刘翔更新社交账号晒出近照 时隔473天更新动态!
2月20日凌晨2点,奥运冠军刘翔更新社交账号晒
资讯
奥运冠军刘翔更新社交账号晒出近照 时隔473天更新动态!
2月20日凌晨2点,奥运冠军刘翔更新社交账号晒
资讯
-
 一个“江浙沪人家的孩子已经不卷学习了”的新闻引发议论纷纷
星标★
来源:桌子的生活观(ID:zzdshg)
没
资讯
一个“江浙沪人家的孩子已经不卷学习了”的新闻引发议论纷纷
星标★
来源:桌子的生活观(ID:zzdshg)
没
资讯
-
 男子“机闹”后航班取消,同机旅客准备集体起诉
1月4日,一男子大闹飞机致航班取消的新闻登上
资讯
男子“机闹”后航班取消,同机旅客准备集体起诉
1月4日,一男子大闹飞机致航班取消的新闻登上
资讯
-
 王自如被强制执行3383万
据中国执行信息公开网消息,近期,王自如新增一
资讯
王自如被强制执行3383万
据中国执行信息公开网消息,近期,王自如新增一
资讯
-
 大家一起关注新疆乌什7.1级地震救援见闻
看到热气腾腾的抓饭马上就要出锅、村里大家
资讯
大家一起关注新疆乌什7.1级地震救援见闻
看到热气腾腾的抓饭马上就要出锅、村里大家
资讯
-
 中央气象台连发四则气象灾害预警
暴雪橙色预警+冰冻橙色预警+大雾黄色预警+
资讯
中央气象台连发四则气象灾害预警
暴雪橙色预警+冰冻橙色预警+大雾黄色预警+
资讯
-
 新增供热能力3200万平方米 新疆最大热电联产项目开工
昨天(26日),新疆最大的热电联产项目—&md
资讯
新增供热能力3200万平方米 新疆最大热电联产项目开工
昨天(26日),新疆最大的热电联产项目—&md
资讯
-
 海南大学生返校机票贵 有什么好的解决办法吗?
近日,有网友在“人民网领导留言板&rdqu
资讯
海南大学生返校机票贵 有什么好的解决办法吗?
近日,有网友在“人民网领导留言板&rdqu
资讯
-
 周星驰新片《少林女足》在台湾省举办海选,吸引了不少素人和足球爱好者前来参加
周星驰新片《少林女足》在台湾省举办海选,吸
资讯
周星驰新片《少林女足》在台湾省举办海选,吸引了不少素人和足球爱好者前来参加
周星驰新片《少林女足》在台湾省举办海选,吸
资讯

